
Growth Hormone
Tesamorelin
Tesamorelin is a synthetic analog of growth hormone-releasing hormone (GHRH) composed of 44 amino acids. It is studied for its role in stimulating endogenous growth hormone (GH) release via pituitary GHRH receptors. Tesamorelin is used exclusively for controlled scientific research and is not approved for therapeutic or diagnostic use.
- Quantity
- 10mg
- Formula
- C221H366N72O67S
- Mol. Weight
- ~5135.89 g/mol
- Purity
- See COA
Standard $9.99 | Expedited $12.99 | Free standard over $200
Download Certificate of AnalysisTesamorelin — The Stabilized GHRH With Clinical Proof
8 min read · 3 citations · Growth Hormone
Tesamorelin vs Sermorelin — Modified vs Unmodified GHRH
Tesamorelin vs Sermorelin compared: stabilized versus unmodified GHRH signaling, DPP-IV resistance, pulse duration, and clinical context.
Best Growth Hormone Secretagogues for Research
A GH-axis roundup comparing GHRH analogs, GHRPs, combined secretagogues, and downstream growth-factor tools by research goal.
CJC-1295 vs Sermorelin vs Tesamorelin — GHRH Analogs Compared
A three-way comparison of the main GHRH analog strategies: native-like reference, stabilized analog, and longer-acting modification logic.
AOD-9604 — Isolating the Fat-Burning Fragment of Growth Hormone
AOD-9604 growth hormone fragment research overview. Lipolysis, anti-lipogenesis, cartilage research, metabolic safety. PubMed cited.
Product Overview
Tesamorelin is a synthetic GHRH analog consisting of the full 44 amino acids of human GHRH modified with a trans-3-hexenoic acid group at the N-terminus. This modification protects the peptide from DPP-IV enzymatic degradation — the enzyme that rapidly inactivates native GHRH and Sermorelin — resulting in significantly improved metabolic stability and a longer half-life.
Tesamorelin is notable in the OSYRIS catalog for having the most recent FDA clinical history among the GH-related compounds. An FDA-approved pharmaceutical version (Egrifta) exists for a specific clinical indication involving HIV-associated lipodystrophy. The clinical trial data from the Egrifta program provides substantial evidence on Tesamorelin's GH-releasing activity, safety profile, and effects on body composition in human subjects.
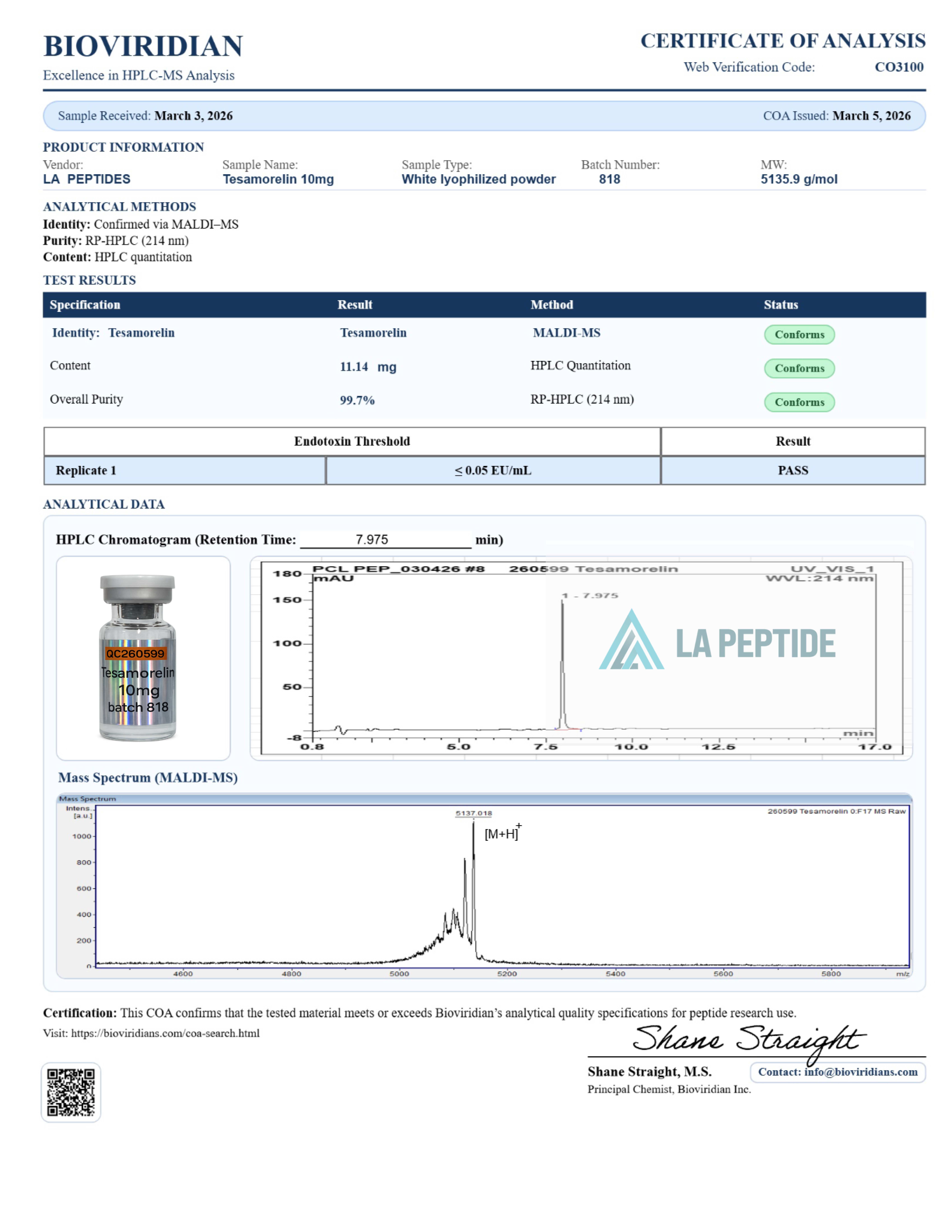
Tesamorelin has a molecular weight of 5135.89 g/mol (C₂₂₁H₃₆₆N₇₂O₆₇S).

Mechanism and Experimental Context
Enhanced GHRH Receptor Agonism ▸
Tesamorelin's DPP-IV-resistant modification gives it a longer window of GHRH receptor activation compared to Sermorelin or native GHRH. This makes it useful for research protocols requiring sustained GHRH-R stimulation rather than the brief pulse produced by unmodified GHRH analogs.¹
Body Composition and Lipodystrophy Research ▸
The clinical trial data from the Egrifta program showed that Tesamorelin reduced visceral adipose tissue (VAT) in HIV-associated lipodystrophy. This body composition data provides a unique evidence base for studying GHRH-mediated effects on fat distribution and adipose tissue biology — a research area where Tesamorelin has more clinical data than any other compound in the GH category.²
Hepatic Steatosis Research ▸
Research has investigated Tesamorelin's effects on liver fat, with clinical data showing reductions in hepatic fat fraction in subjects with both HIV-associated and non-HIV hepatic steatosis. This research connects GH-axis biology to liver metabolism and fatty liver disease models.³
Comparative GHRH Analog Research ▸
Tesamorelin is compared with Sermorelin and CJC-1295 (no DAC) in GH release kinetics studies. The three GHRH analogs have different half-lives and GH release profiles, making comparative research valuable for understanding how peptide modifications affect pharmacological outcomes.
Keep the Research Trail Moving
Source Literature
Teichman SL, et al. "Prolonged stimulation of GH and IGF-I secretion by CJC-1295 in healthy adults." Journal of Clinical Endocrinology & Metabolism, 2006. PubMed: PMID 16352683
Falutz J, et al. "Metabolic effects of a growth hormone-releasing factor in patients with HIV." New England Journal of Medicine, 2007. PubMed: PMID 18003859
Stanley TL, et al. "Effects of tesamorelin on non-alcoholic fatty liver disease." Gut, 2014. PubMed: PMID 23929694
Batch Documentation
Current published batch documentation is surfaced on-page whenever the provider exposes a public COA asset.
Frequently Asked Questions
Questions About Tesamorelin
What is Tesamorelin? ▸
A modified GHRH analog with a trans-3-hexenoic acid group that protects it from enzymatic degradation. It stimulates pituitary GH release with improved stability compared to native GHRH or Sermorelin.
Is Tesamorelin FDA approved? ▸
A pharmaceutical version (Egrifta) is FDA-approved for a specific clinical indication. OSYRIS Tesamorelin is a research-grade compound sold exclusively for laboratory research. It is not a pharmaceutical product.
What is DPP-IV and why does it matter? ▸
DPP-IV (dipeptidyl peptidase IV) is an enzyme that rapidly degrades native GHRH and Sermorelin. Tesamorelin's N-terminal modification blocks DPP-IV cleavage, extending the peptide's active duration in biological systems.
How does Tesamorelin compare to Sermorelin? ▸
Both are GHRH analogs targeting the same receptor. Tesamorelin has greater metabolic stability, resists DPP-IV, and has a longer duration of action. Sermorelin is the simpler, shorter-acting version.
What is lipodystrophy? ▸
Abnormal fat distribution — specifically, the accumulation of visceral abdominal fat. Tesamorelin clinical evidence includes trials showing visceral fat reduction, which makes it uniquely positioned for body composition research in the GH category.
Why does Tesamorelin cross-list to the Metabolic category? ▸
Because its clinical evidence base includes body composition and liver fat data — metabolic endpoints beyond pure GH-axis biology. Its lipodystrophy and hepatic steatosis research connects it to metabolic health models.
How should Tesamorelin be stored? ▸
Lyophilized at -20°C for long-term or 2-8°C for short-term. Protect from light and moisture.
What purity is OSYRIS Tesamorelin? ▸
Independently tested via HPLC and LC-MS. COA downloadable on this page.
Every Batch Tested by an Independent Lab
We publish the Certificate of Analysis for every product. See our full testing process.
All products sold by OSYRIS Health are intended for laboratory research purposes only and are not for human or veterinary use. The information provided on this page describes published scientific research and does not constitute medical advice, diagnosis guidance, or a recommendation for any specific use. Always ensure compliance with local regulations.



